
Radiopharmaceutical Drug Conjugates (RDCs) combine the targeting specificity of biomolecules with the potent therapeutic or diagnostic power of radionuclides, making them a promising frontier in precision oncology. However, the success of RDCs depends on several critical factors, including the stability of chelators, the optimization of linkers, radionuclide selection, and precise in vivo delivery control.
As a global leader in drug delivery technologies, Litchlab specializes in developing high-affinity chelators, multifunctional linkers, and nanocarrier-based delivery systems to optimize RDC performance and accelerate clinical translation.
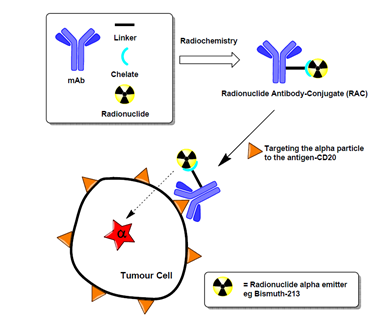
Chelators play a pivotal role in RDC formulations by stabilizing radionuclides, preventing their premature dissociation, and ensuring targeted delivery while minimizing off-target toxicity.
High-affinity chelator library: Includes DOTA, DTPA, TETA, NOTA, HBED, and NETA, optimized for major radionuclides such as ⁶⁸Ga, ⁸⁹Zr, ²²⁵Ac, ¹⁷⁷Lu, ²¹²Pb, and ¹⁶¹Tb.
Custom chelator synthesis: Tailored modifications to enhance hydrophilicity, dissociation kinetics, and biocompatibility for specific RDC applications.
Radiochemical stability evaluation: Utilizing HPLC, LC-MS, and γ-counting to ensure long-term in vivo stability of radiolabeled conjugates.
Scalable production from milligrams to hundreds of grams, supporting both early-stage research and cGMP manufacturing.
Process optimization to maximize yield and purity, ensuring high reproducibility for clinical applications.
Advanced purification strategies, delivering chelators that meet GLP/cGMP quality standards for regulatory compliance.
The linker in RDCs plays a crucial role in controlling release, metabolism, and clearance of the radiopharmaceutical. Litchlab provides cutting-edge linker design and synthesis services, offering cleavable, non-cleavable, metabolically degradable, and biocompatible linkers to optimize RDC behavior in vivo.
Cleavable linkers (disulfide, peptide, ester, pH-responsive): Enable controlled release of radionuclides at target sites.
Non-cleavable linkers (PEG, aliphatic chains): Improve RDC stability and prolong circulation time for enhanced tumor accumulation.
Pharmacokinetically optimized linkers: Adjust lipophilicity/hydrophilicity, biodistribution, and clearance routes to fine-tune RDC efficacy and safety.
| Parameter | Effect |
|---|---|
| Increased lipophilicity | Enhances tissue penetration and cellular uptake |
| Controlled cellular uptake | Extends circulation half-life and improves tumor accumulation |
| Pharmacokinetic modulation | Reduces off-target accumulation and systemic toxicity |
| Hydrophilicity tuning | Minimizes aggregation and enhances biocompatibility |
| Tumor-to-normal tissue ratio adjustment | Enhances tumor specificity and reduces side effects |
| Clearance optimization | Improves renal or hepatobiliary excretion profiles |
Litchlab integrates nanotechnology with RDC delivery through lipid nanoparticles (LNPs), polymer nanoparticles, and microsphere carriers, enhancing RDC stability and therapeutic efficacy.
Key Advantages of Nanocarrier-Based RDC Delivery
✅ Improved tumor accumulation: Enhances selective delivery of RDCs to target sites, minimizing off-target exposure.
✅ Extended systemic circulation: Optimizes pharmacokinetics for prolonged radionuclide retention at tumor sites.
✅ Stimuli-responsive release: Enables controlled drug release triggered by pH, enzymes, or temperature.
✅ Reduced accumulation in healthy organs: Decreases exposure in kidneys, liver, and bone marrow to minimize toxicity.
Litchlab’s RDC delivery technology is widely applied in solid tumors, hematologic malignancies, and radiopharmaceutical imaging, supporting global biopharma companies in advancing RDC drug development.
🔹 Precision oncology applications: Optimizing RDC pharmacokinetics to enhance tumor retention while minimizing toxicity.
🔹 Combination therapy strategies: Enabling RDC co-administration with immune checkpoint inhibitors (PD-1/PD-L1), targeted therapies, and chemotherapies for synergistic effects.
🔹 Clinical translation support: Providing an end-to-end solution from preclinical research to cGMP manufacturing, accelerating RDC IND filings and regulatory approvals.
With the rapid advancements in radiopharmaceuticals, Litchlab remains committed to innovating high-performance chelators, smart linkers, and nanocarrier delivery platforms, empowering global pharma companies to develop safer and more effective RDCs for precision oncology.
For more information, please feel free to contact us at:
E-Mail:RD1@Litchlab.com